Receptor Autoradiography
Receptor autoradiography can be used to identify and map binding sites for a labeled drug in a tissue or organ section.
In sections cut from frozen human or untreated animal tissue, receptor autoradiography is used to measure the affinity of an unlabeled drug, by its competition with the binding of a radiolabeled drug to the target receptor. Autoradiography can also be used to indicate off-target binding sites, where binding is observed even in knockout animals, or in regions of tissue where the target receptor is not expressed.
Ex vivo autoradiography, performed on tissue from drug-treated animals, can be used to determine the receptor occupancy of the drug in the tissue at the time of sacrifice of the animal.
For human tissue use, ethics committee approval is required. Gifford Bioscience manages the application process for this and obtains the tissue from an accredited UK tissue bank.
Autoradiography Protocol
Cryostat-cut tissue sections are incubated with the radiolabeled drug until equilibrium is reached. The sections are washed in fresh buffer to remove unbound radiotracer, dried and imaged using quantitative phosphorimaging. Regions-of-interest (ROIs) are then drawn over each imaged section and radioactivity levels measured. Even small regions (<100 µm) can be quantified.
Example Data
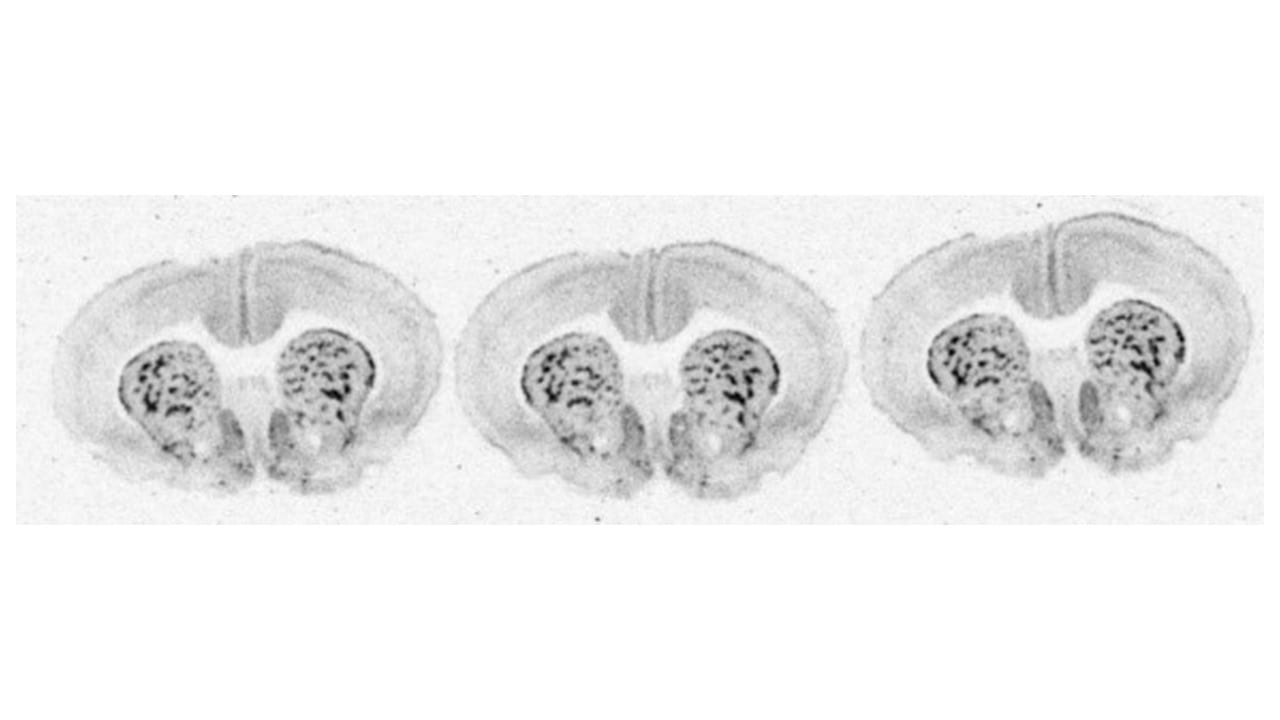
Binding of [3H]DAMGO to µ opioid in coronal sections at the level of the corpus striatum in a rat brain.
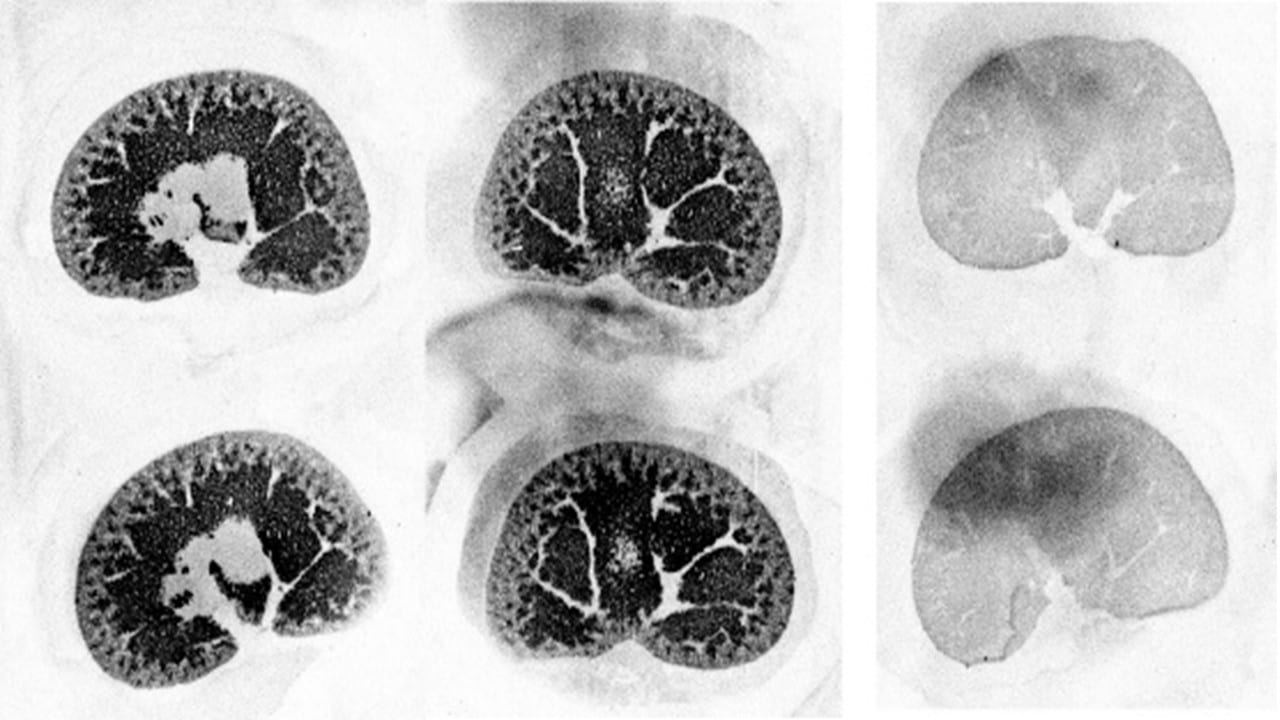
Binding of [3H]LRRK2-IN-1 to the LRRK2 enzyme in rat kidney. Sections in the centre and to the left are total binding. The two sections on the right are non-specific binding, defined using unlabeled LRRK2-IN-1 (10 µM).